|
Видео-ассистированная фенестрация перикарда при фрагментрированных или рецидивных выпотах
Eur J Cardiothorac Surg 1998;14:403��
Video-assisted thoracoscopic pericardial fenestration for loculated or recurrent effusions
Karl Geissbühlera, Alfred Leisera, Jürg Fuhrerb, Hans-Beat Risa
a Department of Thoracic and Cardiovascular Surgery, Inselspital, University of Bern, Bern, Switzerland
b Department of Cardiology, Inselspital, University of Bern, Bern, Switzerland
Corresponding author. Klinik für Thorax- Herz- und Gefässchirurgie, Inselspital, 3010 Bern, Switzerland. Tel.: +41 31 6322330; fax: +41 31 6324036
Abstract
Objective: The validity of video-assisted thoracoscopic pericardial fenestration was prospectively assessed for loculated effusions, effusions previously treated by percutaneous catheter manoeuvres and those with concurrent pleural diseases. Methods: Inclusion criteria consisted of echocardiographically documented pericardial effusions requiring diagnosis or relief of symptoms and recurrent effusions after failed percutaneous drainage and balloon pericardiotomy. Pre-operative CT-scan was used to delineate additional pleural pathology and to determine the side of intervention. All patients were followed clinically and by echocardiographic examination 3 months post-operatively. Results: Twenty-four patients underwent thoracoscopic pericardial fenestration with 11 patients (54%) being previously treated by percutaneous catheter drainage, balloon pericardiotomy or subxyphoidal fenestration. Pre-operative echocardiography revealed septation and loculation in 18 patients (72%). Additional pleural pathology was identified on CT scan in 12 patients (50%) and talc pleurodesis was performed in six patients, all suffering from malignant pleural effusion. The mean operation time was 45 min (range 30㫔 min) with no complications being observed. All patients were followed 3 months post-operatively by clinical and echocardiographic examination; relief of symptoms was achieved in all patients but echocardiography showed a recurrence in one patient (4%). Another recurrence was found by echocardiography after a mean follow-up time of 33 months in the 12 patients suffering from a non-malignant pericardial effusion. No recurrence of pleural or pericardial effusion was observed in the subset of patients with talc pleurodesis. Conclusion: Video-assisted thoracoscopic pericardial fenestration is safe and effective for loculated pericardial effusions previously treated by percutaneous drainage manoeuvres and those with concomitant pleural disease.
Key Words: Pericardium • Window • Thoracoscopy • Recurrence
Chronic or recurrent pericardial effusions are often related to underlying malignant disease such as lung and breast cancer and malignant lymphoma [1]. However, 50% of pericardial effusions in patients with known malignancies are benign. Survival is significantly better in these patients than in those with malignant effusions. Therefore the use of tissue diagnosis in these situations is essential [2]. Pericardial effusions may be related to other aetiologies requiring histological or bacteriological verification such as infections, post-irradiation sequelae, uraemia, collagen vascular diseases and myocardial infarction. In a substantial number of patients, concurrent pleural pathology is present requiring the establishment of simultaneous pleuropericardial diagnosis [3] [4] [5] [6]. Approximately half of the patients suffering from pericardial effusions present with symptoms of cardiac tamponade [7] [8]. In these situations relief of symptoms by means of pericardial decompression is required irrespective of the underlying cause.
While percutaneous catheter drainage and balloon pericardiotomy are increasingly performed for diagnostic and therapeutic purposes, recurrent or loculated effusions are best managed surgically with a pericardial window [2]. Video-assisted thoracoscopic pericardial fenestration has demonstrated usefulness in this respect since this technique affords the opportunity to create a pericardial window of sufficient size with simultaneous assessment of concomitant pleural pathology [3] [4] [5] [6] [9] [10] [11] [12].
This study focuses on video-assisted thoracoscopic pericardial fenestration for loculated effusions and for those previously treated by percutaneous catheter manoeuvres.
Patients and methods
Since 1992, video-assisted thoracoscopic pericardial fenestration has been offered at our institution to patients with recurrent or symptomatic pericardial effusions and with effusions of unknown origin. Inclusion criteria for this study consisted of echocardiographically documented pericardial effusions requiring further diagnosis or relief of symptoms of tamponade and persistent or recurrent effusions after percutaneous drainage and balloon pericardiotomy. Exclusion criteria consisted of echocardiographic or radiological signs of constrictive pericarditis, and pericardial tamponade due to previous cardiac surgery or blunt and penetrating chest trauma.
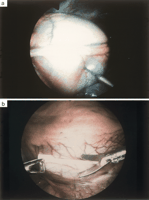
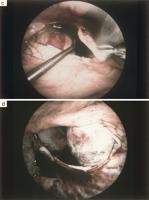
A CT scan was performed in order to identify additional pleural pathology and to determine the side of intervention. The site of intervention was based upon the predominant localization of the loculated perdicardial effusions, and concomitant findings at the pleura. According to the general condition and comorbidity of the patients, the procedure was performed either in the supine position under local anaesthesia (bupivacaine 0.5%) combined with intravenous sedation or in the lateral decubitus under general anaesthesia and double lumen intubation. The scope was inserted via a 5 mm trocar through the fifth intercostal space at the anterior axillary line and two additional 5 mm thoracoports were placed in the sixth intercostal space. Any concomitant pleural effusion was evacuated and pleural pathology was biopsied. A pericardiocentesis was performed under direct vision and the fluid collected for cytological and microbiological analysis ( Fig. 1 a). The pericardium was grasped with a thoracoscopic Duval forceps and incised with curved thoracoscopic Metzenbaum scissors ( Fig. 1b). Loculations and septa were broken and the heart circumferentially freed with a thoracoscopic Senning suction device ( Fig. 1c). A pericardial window was created with a minimum size of 4x4 cm while carefully protecting the phrenic nerve ( Fig. 1d). Due to the different course of the phrenic nerves with respect to both chest cavities, the windows were always performed dorsal to the nerve on the left side and anterior to the nerve on the right side. The pericardial specimen was sent for histological and microbiological analysis. In the event of a combined malignant pericardial and pleural effusion, talc was applied under direct vision on the pleural dome, the chest wall and the diaphragm but not on the mediastinal part of the chest cavity. A chest tube was inserted though one of the thoracoport sites into the chest cavity with no attempt to drain the pericardium.
Fig. 1. Video-assisted thoracoscopic pericardial fenestration performed on the left side: the phrenic nerve is visualized and pericardiocentesis is performed under direct vision, (a); the pericardium is grasped with a Duval forceps and incised with Metzenbaum scissors, (b); fibrinous septa and loculations are broken under direct vision and the heart is freed by use of a thoracoscopic Senning suction, (c); a pericardial window of at least 4x4 cm in size is created, (d).
The patient was extubated and transferred to the intermediate care unit for a few hours of observation, then to the ward on the same day. Chest tubes were removed when the amount of daily drainage was less than 100 ml. A single dose of cephalosporin prophylaxis was given at the beginning of the operation. All patients received patient-controlled analgesia (PCA) post-operatively.
Post-operative morbidity and duration of chest tube drainage and hospitalization were noted. All patients were followed clinically and by echocardiographical examination 3 months post-operatively in order to delineate the development of recurrent effusions. Eleven of the 12 patients with non-malignant disease underwent a second echocardiographical examination during follow-up, with a mean follow-up time of 33 months (range 16㫔 months) after operation.
Results
Twenty-four patients underwent thoracoscopic pericardial fenestration between 1992 and 1996. There were 15 women and nine men with a mean age at operation of 53.6 years, ranging from 18㫬 years. Twelve patients (50%) had undergone percutaneous catheter drainage and balloon pericardiotomy and one (4%) subxyphoidal fenestration prior to thoracoscopic fenestration. The maximum time interval between percutaneous intervention and thoracoscopy was 14 days. Pre-operative echocardiography revealed septations and loculations in 18 patients (72%). Additional pleural pathology was identified in 12 patients (50%) on CT scan and consisted of effusions and pleural thickening or irregularities. Sixteen operations (66%) were performed on the right and eight (34%) on the left side. Double lumen intubation was used in 21 (87%) and local anaesthesia with intravenous sedation in three (13%) patients. In the latter group of patients, the operations could be accomplished under local anaesthesia without complications. Histological, cytological and microbiological examinations of the samples revealed inflammation in eight patients (33%), malignancy in seven (29%), uraemia in four (17%) and post-irradiation sequelae in three (13%). Inflammation, the most common cause, however, was non-specific with no micro-organisms identified, therefore was presumably viral. Additional talc pleurodesis was performed in six patients (25%) suffering from a malignant pleural effusion previously diagnosed by use of percutaneous interventions.
The mean operation time was 45 min and ranged from 30 to 60 min. In one patient an antero-lateral conversion thoracotomy was required to perform pericardectomy due to a thickened and fibrosed pericardium which was underestimated on a pre-operative CT-scan. In all other patients the procedure could be accomplished by thoracoscopy without intraoperative complications being observed. In particular, no bleeding complications occurred despite the fact that no hemostasis was performed in the vicinity of the phrenic nerves. The mean duration of post-operative chest tube drainage and hospitalization were 3.3 days and 10.4 days, ranging from 1 to 7 days, and 4 to 33 days, respectively. Hospitalization times of more than 5 days were related to the underlying disease (malignancy, uraemia) rather than to the procedure per se. Post-operative complications were noted in three patients (12%) and consisted of peripheral pulmonary embolism, vertigo due to a minor cerebrovascular event and supraventricular arrhythmia related to a chest tube which moved into the pericardium. These arrhythmias resolved spontaneously after removal of the drain. There was no treatment-related 30-day mortality but two patients (8%) died due to progression of malignancy. Follow-up 3 months after the operation revealed relief of symptoms in all surviving patients but echocardiography showed a recurrent pericardial effusion in one patient (4%) suffering from viral infection which was asymptomatic and without hemodynamic significance. No recurrence was observed in patients having undergone talc pleurodesis for concomitant malignant pleural effusion. Eleven of the 12 patients with non-malignant disease underwent a second echocardiographical examination during follow-up, with a mean follow-up time of 33 months (range 16㫔 months) after operation. One of these patient showed an asymptomatic recurrent pericardial effusion of 10 mm.
Discussion
Various approaches have been described for diagnostic and therapeutic assessment of pericardial diseases, including pericardiocentesis, percutaneous catheter drainage and balloon pericardiotomy, subxyphoidal pericardial drainage, pericardioperitoneal shunt, subxyphoidal pericardial fenestration and pericardial window via anterior thoracotomy or by means of thoracoscopy. Although there is agreement that constrictive pericarditis is best treated by pericardectomy through a sternotomy, the selection of the optimal drainage procedure for non-constricting effusions is controversial and may vary according to the particular needs and circumstances of the patients [2]. Pericardiocentesis is non-invasive and may result in prompt relief in patients with pericardial tamponade. However, a high complication rate has been reported, especially if pericardiocentesis used for small, loculated effusions, particularly without echocardiographic visualization [13]. Moreover, pericardiocentesis is of limited value for treating thickened fibrinous effusions or a hemopericardium and to establish diagnosis of effusions of unknown origin. Percutaneous catheter drainage is as non-invasive as pericardiocentesis but yields a higher rate of permanent symptom relief since the catheter can be left in place for several days [8]. However, catheter occlusion may occur, and the risks and limitations of this technique are similar to those observed for pericardiocentesis. To overcome these drawbacks, pericardial windows have been recently created by the use of percutaneous balloon pericardiotomy [14]. Subxyphoidal pericardial drainage affords the opportunity to obtain histological samples and to create a larger window, and allows digital exploration in order to break up adhesions and permit more effective drainage of blood and fibrinous debris. It is a quick and inexpensive procedure with a low morbidity and mortality and a recurrence rate as low as 2.6% [15]. However, subxyphoidal pericardial fenestration does not allow direct visual exploration of the pericardial cavity. This might adversely influence the results in patients with prior catheter drainage procedures, which are used with increasing frequency as first line treatments for pericardial effusions. In addition, concomitant pleural pathology is not accessible in the subxyphoidal approach. For these situations, pericardial fenestration via anterior thoracotomy has been advocated to obtain simultaneous access to the pericardium and the pleural cavity [16]. However, this approach is associated with a higher postoperative morbidity and a slower recovery than the subxyphoidal approach [17].
Recently, pericardial fenestration by use of video-assisted thoracoscopy has been proposed in order to gain simultaneous pleuropericardial access and to treat loculated pericardial effusions under direct vision [3] [4] [5] [6] [9] [10] [11] [12]. Our results support the view that this technique is well-suited for this purpose. Video-assisted thoracoscopic fenestration was performed under local anaesthesia and i.v. sedation in four patients (13%). Although all operations could be accomplished by use of local anaesthesia in the supine position and spontaneous breathing, careful inspection of the chest cavity and the creation of a pericardial window of sufficient size (16) was clearly easier to perform under general anaesthesia with exclusion of the lung involved. The operation time (mean 45 min) compares favourably with that of subxyphoidal drainage and no expensive single use materials are required. No wound infection was observed in our patients. Thoracoscopy affords the opportunity to gain simultaneous access to both pleural and pericardial cavities. Fifty percent of our patients were found to have additional pathology of one pleural cavity on CT scan which was accessed at the same time as the pericardial pathology. In half of these patients a malignant pleural effusion was found which was treated by talc pleurodesis after completing pericardial fenestration. In this group there was no recurrence of pericardial or pleural effusion noted on radiographic and echocardiographic assessment 3 months post-operatively. Moreover, thoracoscopic pericardial fenestration allows access to loculated effusions with fibrinous adhesions under direct vision. Loculated fibrinous effusions may occur after percutaneous catheter drainage or balloon pericardiotomy, and may be symptomatic, especially on the right side. 12 patients (50%) had undergone percutaneous catheter drainage with or without balloon pericardiotomy and one (4%) had had a subxyphoidal fenestration prior to thoracoscopic fenestration. Loculated effusions may be approached by thoracoscopy either from the right or from the left side according to the findings on CT-scan. Despite the previous procedures no intra-operative morbidity occurred. Two recurrent pericardial effusions were noted during follow-up, both of them were asymptomatic and were only detected by use of echocardiography. Our findings endorse the need for a careful echocardiographic follow-up in order to detect the true recurrence rate after pericardial drainage procedures.
Video-assisted thoracoscopic pericardial fenestration is safe and effective, especially for loculated effusions, recurrent effusions after previously performed percutaneous catheter drainage manoeuvres and effusions with concomitant pleural disease.
References
Parks J.S., Rentschler R., Wilbur E. Surgical management of pericardial effusion in patients with malignancies. Cancer 1991;67:76㫨.[Medline]
Moores D.W. O., Dziuban S.W. Pericardial drainage procedures. Chest Surg Clin N Am 1995;5:359��.[Medline]
Mack M.J., Landreneau R.J., Hazelrigg S.R., Acuff T.E. Video-thoracoscopic management of benign and malignant pericardial effusions. Chest 1993;103:390��.[Medline]
Liu H.P., Chang C.H., Lin P.J., Hsieh H.C., Chang J.P., Hsieh M.J. Thoracoscopic management of effusive pericardial disease: indications and technique. Ann Thorac Surg 1994;58:1695?.[Abstract]
Nataf P., Jault F., Pouzet B., Dorent R., Lima L., Vaissier E., Benarim S., Levasseur J.P., Delcourt A., Pavie A., Gandjbakhch I. Video-chirurgie des epanchements pericardiques. Technique et resultats. Arch Mal Coeur Vaiss 1996;89:223��.[Medline]
Robles R., Pinero A., Lujan J.A., Fernandez J.A., Torralba J.A., Acosta F., Villegas M., Parrilla P. Thoracoscopic partial pericardiectomy in the diagnosis and management of pericardial effusion. Surg Endosc 1997;11:253��.[Medline]
Press O.W., Liwingston R. Management of malignant pericardial effusion and tamponade. J Am Med Assoc 1987;257:1088.[Medline]
Vaitkus P.T., Herrmann H.C., LeWinter M.M. Treatment of malignant pericardial effusion. J Am Med Assoc 1994;272:59.[Abstract]
Weder W. Thorakoskopische Chirurgie: Indikationen heute. Schweiz Rundsch Med Prax 1993;82:551��.[Medline]
Inderbitzi R., Furrer M., Leupi F. Pericardial biopsy and fenestration. Eur Heart J 1993;14:135��.[Abstract]
Canto A., Guijarro R., Arnau A., Fernandez-Centeno A., Ciscar M.A., Galbis J., Garcia-Vilanova A. Thoracoscopic pericardial fenestration: diagnostic and therapeutic aspects. Thorax 1993;48:1178?.[Abstract]
Furrer M., Hopf M., Ris H.B. Isolated primary chylopericardium: treatment by thoracoscopic duct ligation and pericardial fenestration. J Thorac Cardiovasc Surg 1996;112:1120?.[Free Full Text]
Wong B., Murphy J., Chang C.J. The risk of pericardiocentesis. Am J Cardiol 1979;44:1110.[Medline]
Palacios I.F., Tuczu E.M., Ziskind A.A. Percutaneous balloon pericardial window for patients with malignant pericardial effusion and tamponade. Cathet Cardiovasc Diagn 1991;22:244.[Medline]
Moores D.W. O., Allen K.B., Faber L.P. Subxyphoid pericardial drainage for pericardial tamponade. J Thorac Cardiovasc Surg 1995;109:546.[Abstract/Free Full Text]
Piehler J.M., Pluth J.R., Schaff N.V., Danielson G.K., Orszulak T.A., Puga F.J. Surgical management of effusive pericardial disease. J Thorac Cardiovasc Surg 1985;90:506��.[Abstract]
Naunheim K.S., Kesler K.A., Fiore A.C. Pericardial drainage: subxyphoid versus transthoracic approach. Eur J Cardio-thorac Surg 1991;5:99��.[Abstract]
|